- Apr 15
Cortico Cortical Evoked Potentials: Tracing the Brain’s Hidden Pathways
- Zainab Gangardiwala
- ionm, neuromonitoring, communication, Blogs, speech
- 0 comments

Imagine the brain as a vast web of interconnected pathways, where a single spark in one region can ripple across the entire network. Now imagine being able to watch this communication unfold in real time, seeing not just where activity occurs, but how one region actively influences another. This is the frontier of modern neuroscience, where understanding brain function extends beyond structure and simple activity patterns into the realm of effective connectivity, the cause-and-effect relationships that define neural networks. Brain connectivity is broadly categorized into structural, functional, and effective domains, with effective connectivity capturing the dynamic interactions between regions. One of the most powerful tools for exploring this hidden communication system is Cortico-Cortical Evoked Potentials (CCEPs), an innovative technique that enables clinicians and researchers to trace the brain’s internal pathways with remarkable precision. By briefly stimulating one area of the cortex and recording how signals propagate across connected regions, CCEPs provide a unique window into the brain’s dynamic wiring, which is shaping our understanding of disorders such as epilepsy and guiding safer, more effective brain surgeries.
Cortico-Cortical Evoked Potential (CCEP) stimulation is an invasive technique that uses low-frequency (<2 Hz) electrical stimulation to assess how activation in one brain region elicits responses in connected areas. By mapping these responses, CCEP enables reliable, real-time localization of functionally related brain regions and has been widely used to delineate functional networks, including the human language network, limbic system, and motor pathways. Clinically, CCEPs are most commonly applied to identify the origin and spread of seizures in patients with severe, refractory epilepsy. Increasing evidence suggests that seizures arise from disturbances within distributed neural networks rather than a single isolated focus; therefore, traditional modalities such as EEG and MRI are often complemented by CCEP to more precisely identify regions of epileptogenicity and pathways of seizure propagation within the brain.
As mentioned, CCEP is an invasive technique that involves electrical stimulation directly on the cortical surface of the brain. To access the awake human brain, patients with severe, medically intractable epilepsy who are undergoing surgical evaluation are recruited for research. CCEPs are typically recorded after clinical seizures have been documented and baseline doses of antiepileptic medications have been reinstated. EEG is performed prior to implantation to identify the hemisphere and lobe generating the seizures; however, precise localization of epileptic activity requires direct electrode contact with the cortex, where CCEP becomes particularly useful.
There are two main techniques for intracranial monitoring: electrocorticography (ECoG), which involves placing strips or sheets of electrodes on the cortical surface, and stereo electroencephalography (SEEG), which involves penetrating the brain with electrodes. CCEP mapping begins with the injection of current between a pair of adjacent electrodes, with brief stimulation of up to 10 mA typically being tolerated. This stimulation triggers electrical responses in the targeted region and in adjacent or remote areas connected to it. CCEPs typically consist of an early N1 wave, reflecting direct pathways, and a later N2 wave, reflecting polysynaptic or
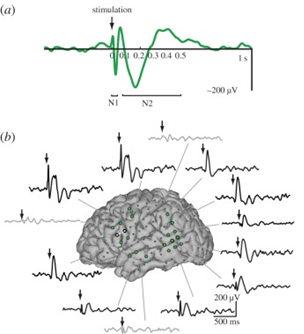
Figure: a) CCEP responses include the early N1 and late N2. (b) Bipolar stimulation is applied between adjacent electrodes (dotted white lines), and significant and non-significant CCEPs (green and gray colored electrodes, respectively) are measured. Examples of CCEP waveforms are shown in several significant (black) and non-significant (gray) regions (Keller et al.).
Several studies have employed CCEP to identify potential biomarkers that may define the epileptogenic zone (EZ), and CCEPs are often used clinically in intraoperative brain mapping to preserve essential functional areas during epilepsy or tumor surgeries. One review indicated that individuals who underwent complete resection of areas showing abnormal CCEP waveforms demonstrated significantly better outcomes (Qiang et al., 2025). CCEP is superior to non-invasive imaging techniques, such as MRI or EEG, which indirectly or remotely measure neural activity, because CCEP directly provides in vivo measurements of brain dynamics with high spatial and temporal resolution. Furthermore, in patients already undergoing invasive brain surgery, CCEP mapping is a feasible technique that requires minimal modification to existing invasive electrical neuromonitoring procedures.
One of the biggest limitations of CCEP is its invasive nature, restricting its use primarily to patients with refractory epilepsy, tumors, or other cortical pathologies who are undergoing surgical intervention. This limits the ability to examine effective connectivity in individuals with normal brain function. Additionally, although CCEPs are commonly used in both clinical and research settings, there is no consistent standard for measuring or defining CCEP responses, and significant variability in detection methods persists across studies. This lack of standardization can lead to missed detections or misclassification of responses as artifacts.
Overall, Cortico-Cortical Evoked Potentials (CCEP) are a powerful tool for measuring effective connectivity in the human brain, offering direct insight into how neural regions interact. As research advances, future directions for CCEP include developing standardized protocols for stimulation and analysis, enhancing its reliability, and expanding its role in intraoperative settings. By improving seizure localization accuracy and informing surgical planning, CCEP has the potential to significantly enhance patient outcomes while deepening our understanding of large-scale brain network organization. Beyond simply mapping the brain, CCEPs provide a more profound view of how the brain functions as an interconnected system, revealing the pathways through which signals travel and interact. This network-based perspective is shifting clinical practice from focusing on isolated regions to understanding dynamic interactions across the brain. Although challenges such as invasiveness and methodological variability remain, ongoing advancements continue to refine this technique. As the field progresses, CCEPs hold promise not only for improving surgical outcomes in patients with epilepsy and brain tumors but also for uncovering fundamental principles of brain organization, bringing us closer to decoding the brain’s most complex and hidden connections.
About the Author:
Zainab Gangardiwala is a medical student at the Texas Tech University Health Sciences Center. She is passionate about neuroscience and hopes to specialize in a related field. She is also a research intern at Global Innervation LLC in Dallas, Texas.
References:
Al-Sadek, Tamara, et al. “Methodologies to Detect Cortico-Cortical Evoked Potentials: A Systematic Review.” Frontiers in Human Neuroscience, vol. 19, 1 Sept. 2025, https://doi.org/10.3389/fnhum.2025.1636115. Accessed 29 Nov. 2025.
Global Innervation LLC. (2025). Global Innervation: Innervate your world. Retrieved April 15, 2026, from https://www.globalinnervation.com/
Keller, Corey J, et al. “Mapping Human Brain Networks with Cortico-Cortical Evoked Potentials.” Philosophical Transactions of the Royal Society B, vol. 369, no. 1653, 5 Oct. 2014, pp. 20130528–20130528, https://doi.org/10.1098/rstb.2013.0528.
KUNIEDA, Takeharu, et al. “New Approach for Exploring Cerebral Functional Connectivity: Review of Cortico-Cortical Evoked Potential.” Neurologia Medico-Chirurgica, vol. 55, no. 5, 2015, pp. 374–382, https://doi.org/10.2176/nmc.ra.2014-0388. Accessed 18 Feb. 2020.
Lega, Bradley, et al. “Cortico-Cortical Evoked Potentials for Sites of Early versus Late Seizure Spread in Stereoelectroencephalography.” Epilepsy Research, vol. 115, 1 Sept. 2015, pp. 17–29, www.sciencedirect.com/science/article/pii/S0920121115000820?via%3Dihub, https://doi.org/10.1016/j.eplepsyres.2015.04.009.
Lhatoo, Samden D, et al. Invasive Studies of the Human Epileptic Brain: Principles and Practice. Oxford, UK, Oxford University Press, 2019.
Matsumoto, R., et al. “Functional Connectivity in Human Cortical Motor System: A Cortico-Cortical Evoked Potential Study.” Brain, vol. 130, no. 1, 21 Nov. 2006, pp. 181–197, https://doi.org/10.1093/brain/awl257. Accessed 18 Feb. 2020.
Matsumoto, Riki, et al. “Functional Connectivity in the Human Language System: A Cortico-Cortical Evoked Potential Study.” Brain, vol. 127, no. 10, 25 July 2004, pp. 2316–2330, https://doi.org/10.1093/brain/awh246. Accessed 18 Feb. 2020.
Prime, David, et al. “Considerations in Performing and Analyzing the Responses of Cortico-Cortical Evoked Potentials in Stereo-EEG.” Epilepsia, vol. 59, no. 1, 16 Nov. 2017, pp. 16–26, https://doi.org/10.1111/epi.13939.
Qiang, Zekai, et al. “Cortico‐Cortical Evoked Potentials: Analytical Techniques and Emerging Paradigms for Epileptogenic Zone Localization.” Epilepsia, vol. 66, no. 9, 23 May 2025, pp. 3087–3104, https://doi.org/10.1111/epi.18467. Accessed 6 Apr. 2026.