- May 14, 2025
Optimizing Cranial Nerve Protection: Multimodality IONM in High-Risk Brain Surgeries
- Faisal Jahangiri
- 0 comments
Introduction
The human body contains 12 pairs of cranial nerves (CN), all of which can be monitored intraoperatively except for the olfactory nerve (CN I). In high-risk brain surgeries, multimodal intraoperative neurophysiological monitoring (IONM) plays a crucial role in preserving cranial nerve function and preventing post-operative neurological deficits.
Various IONM modalities provide real-time feedback, aiding surgeons in nerve identification, mapping, and functional assessment. These modalities include:
· Electromyography (EMG): Spontaneous (s-EMG) and triggered (t-EMG) monitoring (Fig. 1).
· Motor Evoked Potentials (MEP): Cranial Nerve MEP (CN-MEP) for corticobulbar tract evaluation (Fig. 2).
· Nerve Action Potentials (NAP): Assessing peripheral nerve function.
· Visual Evoked Potentials (VEP): Monitoring optic nerve (CN II) integrity (Fig. 3).
· Somatosensory Evoked Potentials (SSEP): Applicable for trigeminal (CN V) and facial (CN VII) nerves.
· Brainstem Auditory Evoked Potentials (BAEP): Essential for vestibulocochlear (CN VIII) nerve monitoring. (Fig. 4).
Cranial Nerve-Specific Monitoring Approaches
Optic Nerve (CN II): As a sensory nerve, CN II can be monitored via visual stimulation (flash goggles) with recordings from the retina (Electroretinography, ERG) and visual cortex (VEP).
Trigeminal Nerve (CN V): The sensory division of CN V can be monitored using Trigeminal SSEP (TSEP), providing insight into functional integrity.
Vestibulocochlear Nerve (CN VIII): CN VIII monitoring involves auditory stimulation and multi-level response recordings, including:
· Electrocochleography (ECoch): Capturing responses from the tympanic membrane and cochlea
· Auditory Nerve Compound Action Potentials (AN-CAP): Direct recordings from the auditory nerve
· Brainstem Auditory Evoked Potentials (BAEP): Evaluating brainstem auditory pathway function
BAEP responses can be affected by factors such as nitrous oxide (N₂O), hypothermia, and inhalational anesthetics. These influences must be carefully managed during surgery when hearing preservation is a priority.
Motor Cranial Nerve Monitoring
The remaining cranial nerves CN III (Oculomotor), IV (Trochlear), VI (Abducens), VII (Facial), IX (Glossopharyngeal), X (Vagus), XI (Spinal Accessory), and XII (Hypoglossal) are primarily motor nerves and are monitored using spontaneous EMG (s-EMG) and triggered EMG (t-EMG). These techniques facilitate:
· Real-time nerve function assessment
· Precise nerve localization and mapping
· Immediate feedback for surgical intervention
EMG responses are both visually displayed and audible, allowing surgeons to quickly detect changes and adjust their approach accordingly. Various perioperative factors, such as muscle relaxants, inhalational agents, ischemia, and hypothermia, may influence CN EMG, MEP, and VEP responses.
CN-MEP is particularly valuable for evaluating corticobulbar tract integrity, ensuring proper neural connectivity. The optimal anesthetic regimen for cranial nerve monitoring is Total Intravenous Anesthesia (TIVA) without muscle relaxants.
Intraoperative Stimulations & Insults
During surgery, monopolar and bipolar stimulations assist in nerve identification and mapping:
· Monopolar stimulation: Higher sensitivity but lower specificity
· Bipolar stimulation: Higher specificity but lower sensitivity
IONM detects intraoperative insults, including mechanical, ischemic, electrical, and chemical injuries which could otherwise compromise nerve function.
Conclusion
Multimodal intraoperative neurophysiological monitoring is an essential tool for preventing cranial nerve injuries during high-risk brain surgeries. Optic (CN II) and vestibulocochlear (CN VIII) nerves benefit from visual and auditory response monitoring, ensuring the preservation of vision and hearing. Motor nerves (CN III-XII) can be effectively monitored through EMG techniques, allowing surgeons to make informed decisions regarding tumor resection and surgical manipulation.
By providing real-time feedback, IONM minimizes post-operative neurological deficits and significantly enhances patient safety. A timely IONM alert during surgery allows the surgical team to identify potential issues and take immediate corrective actions, ultimately leading to better patient outcomes and improved functional status of the nervous system.
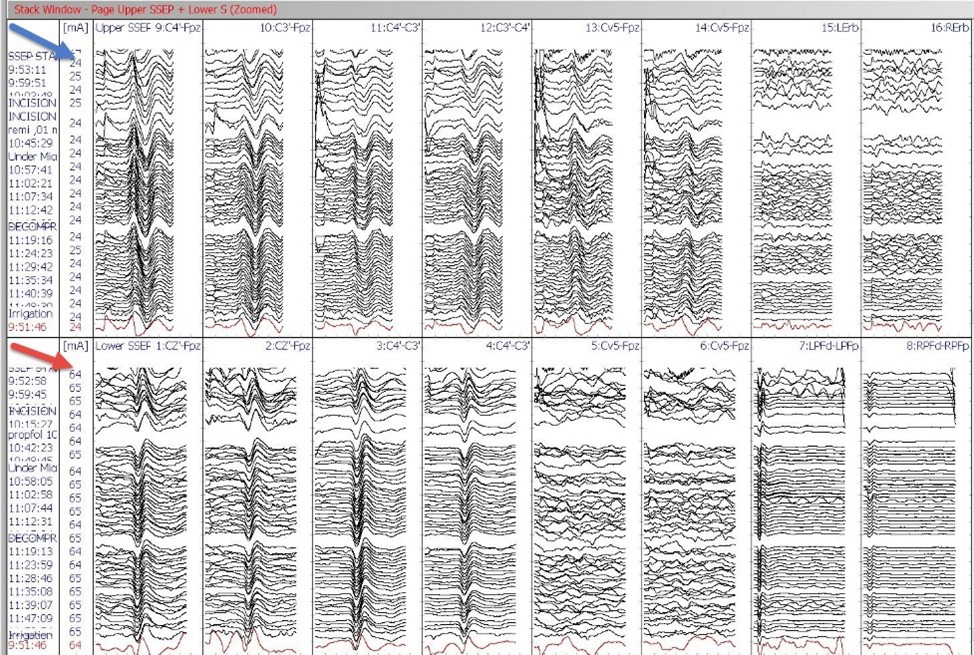
Figure 1: Somatosensory Evoked Potentials (SSEP) stack data bilaterally recorded from scalp, subcortical and peripheral sites by Median nerve stimulation at the wrist (blue arrow) and Posterior Tibial nerve stimulation at medial ankles (red arrow).
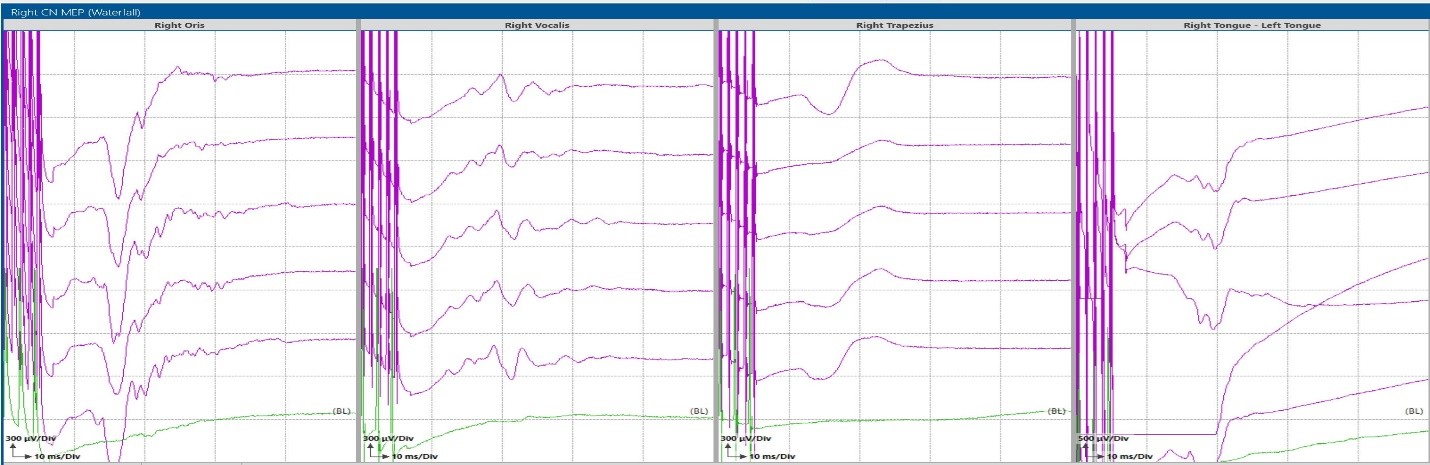
Figure 2: Corticobulbar Transcranial electrical motor Evoked Potentials (TCeMEP) recorded from Oris (CN VII), Vocal Cords (CN X), Trapezius (CN XI), and Tongue (CN XII) muscles during a fourth ventricle tumor resection.
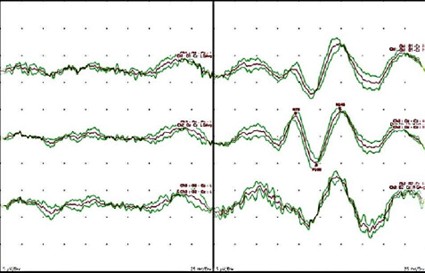
Figure 3: Visual Evoked Potentials (VEP) [Cranial Nerve II]. Left: Absent Flash VEP (FVEP) responses due to loss of vision pre-operatively in the left eye. Right: Reliable FVEP responses present in response to right eye stimulation.
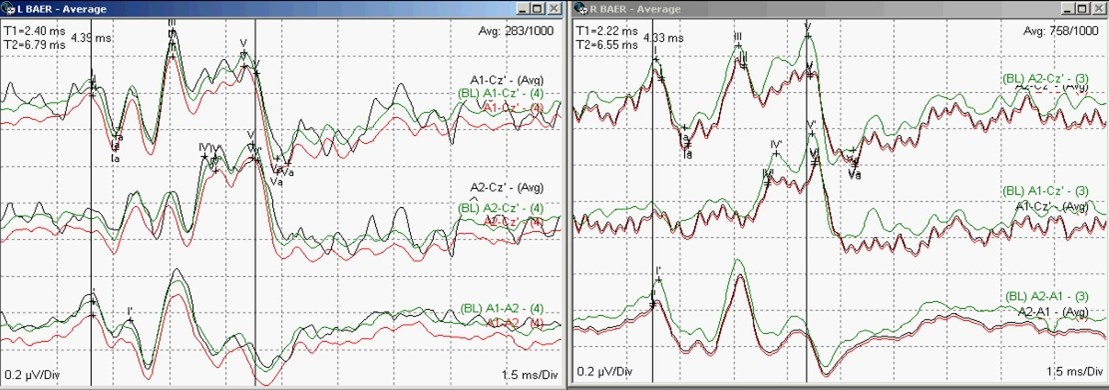
Figure 4: Brainstem Auditory Evoked Potentials (BAEP) [Cranial Nerve VIII] recordings in response to left and ear stimulation (Waves I, III and V present bilaterally).
References
Jahangiri FR, Minhas M, Jane J Jr. Preventing lower cranial nerve injuries during fourth ventricle tumor resection by utilizing intraoperative neurophysiological monitoring. Neurodiagn J. 2012 Dec;52(4):320-32.
Farhan, Rida, et al. Introduction to Neurophysiology, edited by Faisal R Jahangiri, Kendall Hunt Publishing, Dubque, IA, 2023, pp. 191–214.
Nadeem, W., Khattak, W. H., Jahangiri, R. H., Asad, H., Farooq, L., Blaylock, J., Cronce-Solano, L., Garza, R. R., Fell, W. R., Basha, I. S., & Jahangiri, F. R. (2023). Facial Nerve Monitoring During Parotidectomy: A Case Series. J of Neurophysiological Monitoring, 1(1), 1–12. https://doi.org/10.5281/zenodo.8206378